SARS-CoV-2 and the newborn: what do we know? (Pt.2)
| 05 May 2022
0 Replies
DESCRIPTION
Continuing our journey related to SARS-CoV-2 and the placenta, a very interesting contribution came from Facchetti et Al. who in 2020 reported their experience of mothers positive for the virus.
In detail, they analyzed a cohort of 15 women positive for the virus and whose placentas had undergone histopathological analysis from February 7 to May 15, 2020. Among these, the case of a young woman in whom transmission was reported was highlighted vertical (maternal-fetal) of SARS-CoV-2.
The authors, starting from the panorama that emerged in those months, reported that although viral placental infection had already been described in the literature, little was known about the potential vertical transmission of the virus.
Therefore, authors referred to a 29-year-old woman hospitalized at 37 weeks + 5 of the first pregnancy, due to fever (38.0°C) and idiopathic thrombocytopenia, diagnosed at the beginning of pregnancy.
Patient tested positive for SARS-CoV-2 RNA and X-ray imaging showed parenchymal pulmonary thickening. Because of the fever and worsening of thrombocytopenia, induced vaginal delivery was chosen, with support ventilation due to the onset of O2 saturation of 95% in room air. The male infant weighed 2840 kg and presented an APGAR score of 9/10.
Twenty-four hours after delivery the newborn had developed fever, difficulty breathing, vomiting, abdominal distension, hypotonia and mild skin rash. Chest X-ray revealed interstitial pneumonia and was transferred to intensive care, where mechanical ventilation was applied. After the birth test for SARS-CoV-2 the RNA on the nasopharyngeal swab was inconclusive, while it was positive 36 and 72 h after birth and at the age of 17 days. After giving birth, the mother recovered quickly, the fever subsided and the puerperal course proceeded normally.
The child was discharged on day 18 after birth in good general condition. Two months later delivery both mother and child were healthy and asymptomatic.
The patient's monoamniotic-monochorionic placenta weighed 448 gr (25-50th percentiles for age). No significant anomalies were found on umbilical cord insertion, umbilical cord vessels, fetal surface and membranes, while the dark brown chorionic parenchyma contained smaller light areas.
On microscopic examination, the chorionic plate both in the fetal and maternal area showed multiple foci (corresponding to about 40% of the total surface) with intervillous inflammatory infiltrate, composed of numerous monocytoid cells and mature macrophages and neutrophils.
In areas showing inflammation, villi showed variable variations consistent with fetal vascular malperfusion with avascular villi and stroma-vascular cariorressi. The dangerous deposition of fibrin was prominent in many areas.
In the remaining parenchyma, normal villi and focal chorangiosis was observed. No intra-villous inflammatory infiltrate (villite) was evident. Immunostain for SARS-CoV-2 proteins showed positivity in cytoplasm of the syncytiotrophoblast for both protein S and protein N (Fig. 1A and B). Protein N was also expressed by rare intervals macrophages and Hofbauer cells. In particular, while anti-nucleocapsid staining was homogeneously strong throughout the placental parenchyma, protein S immunoreactivity was variable and particularly strong in areas with abundant intervillous inflammation (Fig.1C).
SARS-CoV-2 proteins were not detected in the villi capillaries. Electron microcopy of formalin-fixed placental tissue revealed fine morphologic preservation. On negative stain EM (nsEM), they observed particles morphologically compatible with coronavirus. They consisted of round particles with an external “corona” made of peplomers, and their size ranged between 85 and 90 nm to 110x120 nm (including peplomers). Isolated coronavirus but also couples or small clusters of particles were identified (Fig. 2A).
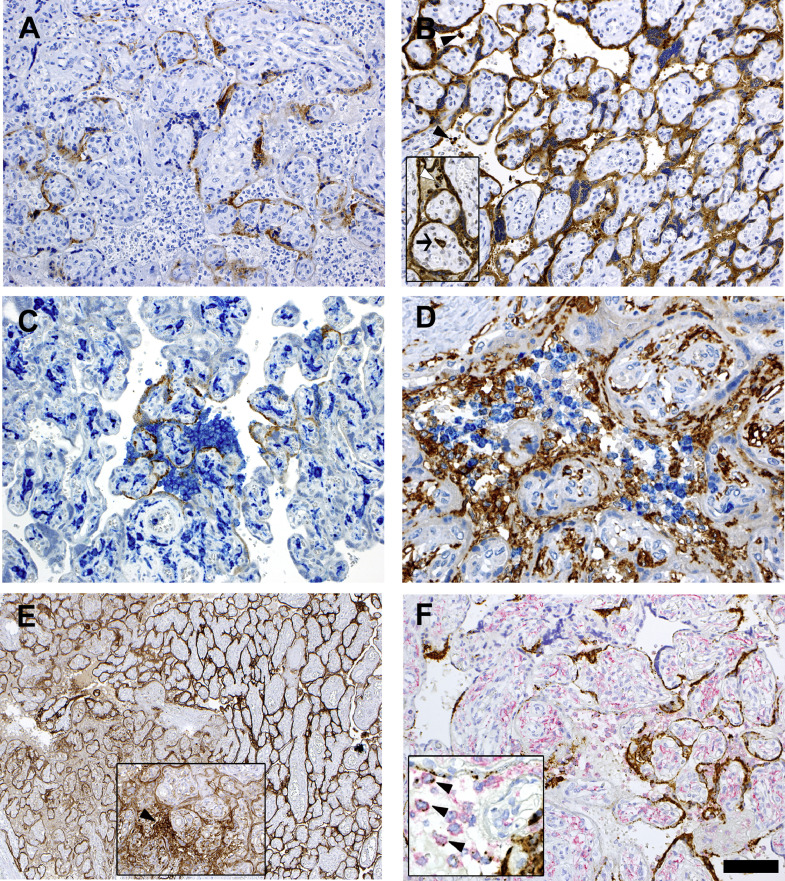
(A, B) Immunostainings for SARS-CoV-2 spike (A) and nucleocapsid (B) proteins. Syncytiotrophoblast is positive for both proteins. Note the more extensive and stronger positivity for nucleocapsid expression, which also localizes to intervillous inflammatory cells (inset, arrowheads) and an Hofbauer cell (inset, arrow).
(C) Double stain for spike protein (brown) and CD14 (blue) indicates higher density of inflammatory infiltrate at sites where the viral protein is more intensely expressed.
(D) Double staining for CD14 (brown) and CD66b (blue) expression, respectively identifying monocyte-macrophages and neutrophils in the inflammatory infiltrate occupying the intervillous spaces. Note the CD14-positive Hofbauer cells in the villi.
(E) PD-L1 staining shows strong expression in the syncytiotrophoblast in areas displaying inflammation (left side). The inset shows an area infiltrated with inflammatory cells, which express PD-L1 (arrow), similarly to intravillous Hofbauer cells.
(F) SARS-CoV-2 RNA in situ hybridization using the S antisense probe (brown stain) combined with anti-CD14 immunostain (red stain). Strong expression of viral RNA is observed in the syncytiotrophoblast, and in scattered CD14+ intervillous mononuclear cells, identified as monocyte-macrophage (inset, arrowheads). (Bar corresponds to 100 μm for figs. A-B-B inset-C-F, 50 μm for fig. D and F inset, 200 μm for fig. E and 150 μm for E inset).

(A, B) Negative stain electron micrograph of particles obtained from a homogenate of formalin-fixed placenta (A, bar 100 nm) and ultrathin sections of free particle in the cytoplasm of a syncytiotrophoblast cell (B, bar 100 nm). In both cases morphological features consistent with coronavirus are observed.
(C) Ultrathin sections of a villous. The right and the left squares include a portion of the syncytiotrophoblast and of a capillary vessel; the respective details are illustrated in fig. C1-C2 and C3-C4. In C2 the arrow points to an aggregate of SARS-Cov-2 nucleocapsid inclusions, the arrowhead to a particle consistent with mature coronavirus. In C4 the arrow points to a particle with the typical morphological features of coronavirus within the endothelium. (C1, bar 2 μm; C2, bar 1 μm; C3, bar 2 μm; C4, bar 250 nm). (D, D1) The endothelium of a fetal capillary (D, bar 5 μm) close to the villus surface shows particles morphologically consistent with SARV-CoV-2 in the cytoplasm (D1, high magnification of the area shown in C, bar 500 nm).
Lascia un commento